Immune checkpoint inhibitors (ICIs) are a new class of anticancer drugs. Because they can block the effect of inhibitory immune checkpoint receptors, ICIs introduce a new era of cancer immunotherapy. Many companies are developing new ICIs and immunomodulators for cancer treatment. However, the T cell-activating capacity of drug candidates in vitro assays has limitations and needs improvement. Creative Biolabs provides a comprehensive range of customized, high-quality immune checkpoint functional assays services such as immune checkpoint mixed lymphocyte reaction (MLR) assay services to support biomedical sciences research communities worldwide.
With abundant experience and advanced platforms, Creative Biolabs has developed some MLR methods based on one-way MLR or two-way MLR and many ways to set them up by changing the types of cells used and choosing appropriate readouts depending on your research question. These assays will enable our clients to understand and monitor any significant immunomodulatory effects of immune checkpoint molecules. Partnering with our specialists will help you progress quickly and make informed decisions for the next step of your project.
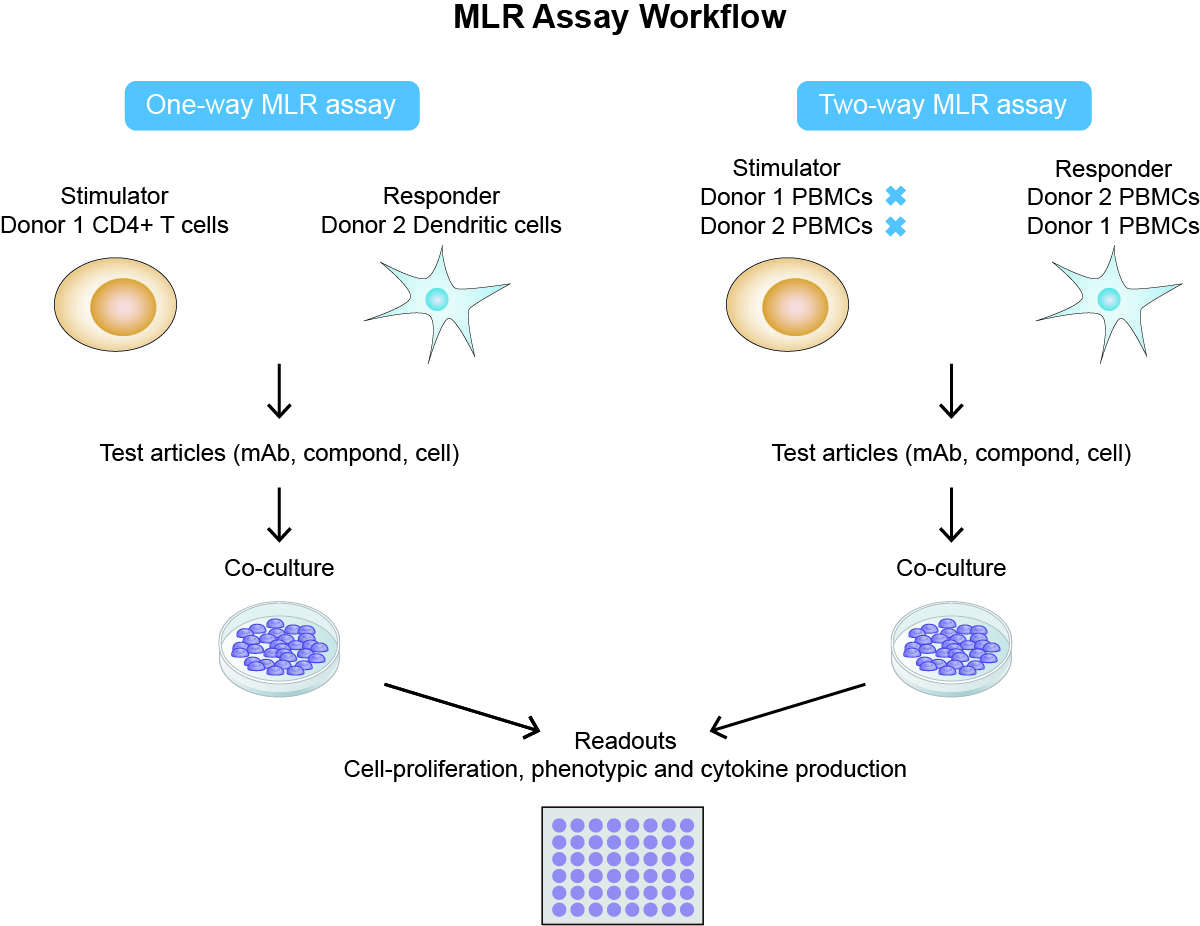 Fig 1. One-way and two-way MLR assay workflow. (Creative Biolabs)
Fig 1. One-way and two-way MLR assay workflow. (Creative Biolabs)
The principle of the MLR is that immune cells from different individuals are co-cultured together to model an immune microenvironment and to trigger the non-self-recognition required for immune checkpoint regulation, resulting in cell activation, DNA synthesis, and proliferation. This is caused by the particularly vigorous allogeneic responses and recognition of an HLA mismatch between two unrelated donors, which provokes an immune response from the T cells.
Using an MLR we can approximate these suppressed immune states (such as a T-cell exhaustion assay) to investigate how we can recover function (immune checkpoints). MLR is crucial for the pre-clinical stages of the therapeutics development pipeline (such as drugs, biomaterials, cells, or other therapeutic products). It is a great assay to help us investigate the potential therapeutic molecule or biologics by evaluating the ability for stimulation/activation of T-cells. It also provides critical information for immunological responsiveness for the mechanism of action, efficacy, and safety of your product.

Creative Biolabs has a suite of MLR readout options tailored to mechanisms of interest for design experiments and will assist with data analysis for your specific requirements.
Proliferation can be simply measured by ELISA using a radioactive marker, usually tritiated thymidine (3H-Tdr), or more recently a non-radioactive marker MTT or bromodeoxyuridine (BrdU) assay. Other markers such as CFSE and EdU can be used for flow cytometry. The quantification of proliferating generations of CD3, CD4, and CD8 positive T cells and activation markers profile (ie. CD209, CD1a, CD80, CD83, and CD86) can be characterized by multiplexed flow cytometry. Additional endpoints such as cytokines can be added to the experimental data sets. Many different cytokines can be tested such as IL-2 and IFNγ (one of the most representative surrogates of T cell activation).
Creative Biolabs strives to retain our customers through competitive cost, strong communication, timeliness, and responsive programs. We are committed to efficiency, confidentiality, rapidity, and accuracy. We desire to work closely with our clients to achieve long-term beneficial relationships. If you want to know more information, please directly contact us.
All listed customized services & products are for research use only, not intended for pharmaceutical, diagnostic, therapeutic, or any in vivo human use.
USA
Tel:
Fax:
Email:
Copyright © 2026 Creative Biolabs. All Rights Reserved.