Considering the immune tolerance in the tumor microenvironment, Creative Biolabs has devoted significant efforts to provide immune checkpoint antibody conjugation services. We strive to retain our customers through competitive cost, strong communication, timeliness, and responsive programs. We desire to work closely with our clients to achieve long-term beneficial relationships.
Immune checkpoints are comprised of multiple inhibitory pathways that involve the interactions of co-receptors and ligands expressed on the surfaces of T lymphocytes (T cells) and antigen-presenting cells. Once a T cell recognizes and binds to its cognate antigen through the T cell receptor (TCR), the interaction exerts a co-stimulatory or inhibitory downstream signaling to either suppress or activate the T cell. Cytotoxic T lymphocyte-associated molecule-4 (CTLA-4) and programmed cell death receptor-1 (PD-1) are the most studied members of this group of cell surface receptors. Immune checkpoint blockade (ICB) therapy has developed in cancer biotherapy.
LAG-3 was discovered in 1990. It is a type I transmembrane protein that is identified on activated human NK and T cell lines. Under inflammatory conditions, LAG-3 is expressed in vivo on the surface of activated CD4+, CD8+, and NK cells. There are currently four LAG-3 modulating agents that have entered the clinic as anti-cancer therapeutics, with several more in preclinical development.
TIM-3 (T Cell Immunoglobulin Mucin 3) is a type I transmembrane protein that is discovered initially to identify novel cell surface molecules that would mark interferon (IFN)-γ-producing T helper type 1 (Th1) and type 1 cytotoxic T (Tc1) cells. TIM-3 plays a key role in inhibiting Th1 responses and the expression of cytokines such as TNF and INF-γ. Dysregulation of TIM-3 expression has been associated with autoimmune diseases.
CD27 is a potent co-stimulatory molecule. It is important for the clonal expansion of naïve T cells and differentiation into effector cytotoxic lymphocytes. Its ligand CD70 molecule is mainly expressed by activated lymphocytes and mediates B cell activation and immunoglobulin production. A humanized agonistic anti-CD27 antibody is also under exploration in phase I/II trials against multiple solid tumor malignancies, including colorectal cancer (CRC), non-small cell lung cancer (NSCLC), ovarian, and head and neck tumors.
Creative Biolabs provides world-class immune checkpoint antibody conjugation services based on the latest antibody engineering technology platforms. Our scientists employ the most advanced techniques to provide comprehensive services to meet each customer's individual requirement for immune checkpoint research and therapeutic markets.
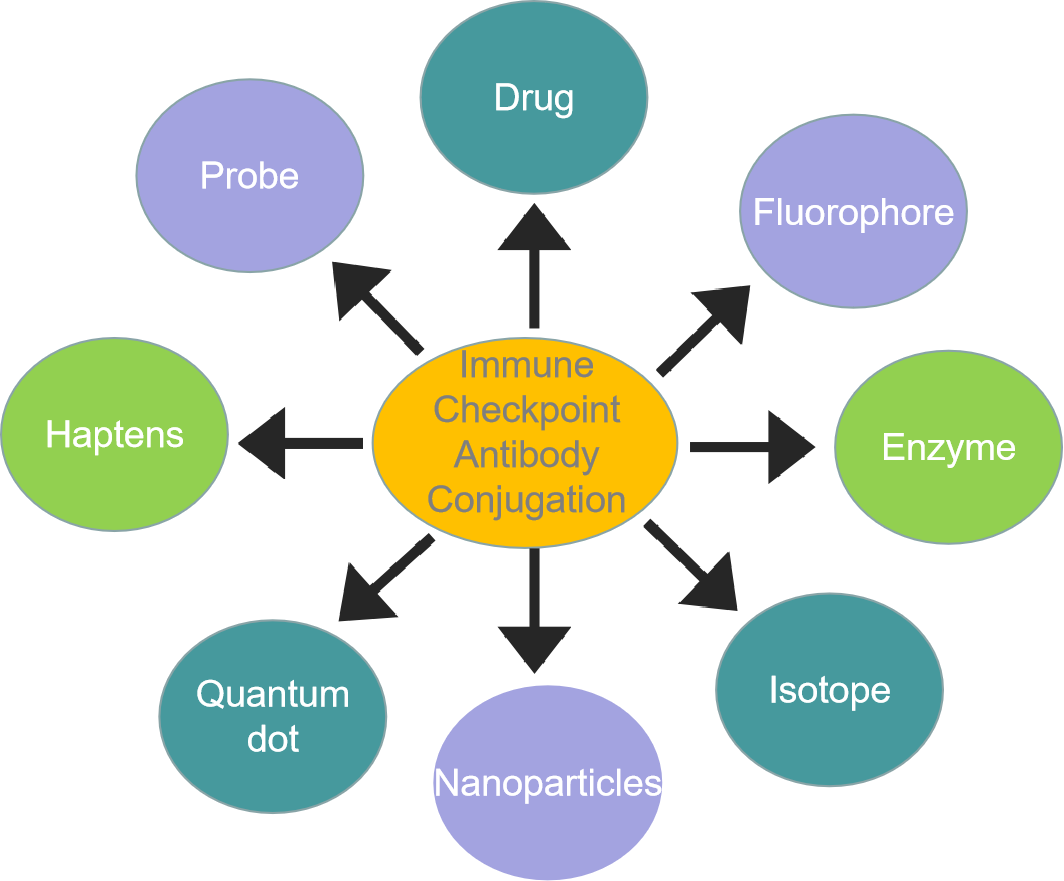
What We Can Do?Creative Biolabs has a management team with an average of 10+ years of industry experience. Our goal is to provide specialized, high-quality immune checkpoint antibody conjugation services to successfully meet the requirements of your trial in the most effective and cost-efficient manner possible. If you want to know more information, please directly contact us.
All listed customized services & products are for research use only, not intended for pharmaceutical, diagnostic, therapeutic, or any in vivo human use.
USA
Tel:
Fax:
Email:
Copyright © 2026 Creative Biolabs. All Rights Reserved.